Abstract
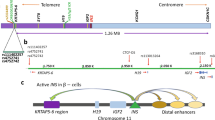
Parental imprinting is a phenomenon in mammals whereby the maternal and paternal alleles of a gene are differentially expressed. Three murine genes have been shown to display this type of allele–specific expression. Two of them, insulin–like growth factor–2 (Igf–2) and H19, map to the distal end of mouse chromosome 7, but are imprinted in opposite directions. Pulsed–field gel electrophoresis and large–fragment DNA cloning were utilized to establish a physical map that includes H19 and Igf–2. Igf–2 lies ∼90 kilobases of DNA 5′ to H19, in the same transcriptional orientation. This physical proximity is conserved in humans, based on pulsed–field gel analysis. We conclude that H19 and Igf–2 constitute an imprinted domain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
McGrath, J. & Solter, D. Inability of mouse blastomere nuclei transferred to enucleated zygotes to support development in vitro. Science 226, 1317–1319 (1984).
Surani, M.A.H., Barton, S.C. & Norris, M.L. Development of reconstituted mouse eggs suggest imprinting of the genome during gametogenesis. Nature 308, 548–550 (1984).
Barton, S.C., Surani, M.A.H. & Norris, M.L. Role of paternal and maternal genomes in mouse development. Nature 311, 374–376 (1984).
Cattanach, B.M. Non-disjunction tests with Robertsonian translocations. Mouse News Lett. 66, 62–63 (1982).
Cattanach, B.M. & Kirk, M. Differential activity of maternally and paternally derived chromosome regions in mice. Nature 315, 496–498 (1985).
Cattanach, B.M. Parental origin effects in mice. J. embrol. exp. Morph. Suppl. 97, 137–150 (1986).
Searle, A.G. & Beechey, C.V. Genome imprinting phenomena on mouse chromosome 7. Genet. Res. 56, 237–244 (1990).
Barlow, D.P., Stoger, R., Herrmann, B.G., Saito, K. & Schweifer, N. The mouse insulin-like growth factor type-2 receptor is imprinted and closely linked to the Tme locus. Nature 349, 84–87 (1991).
DeChiara, T.M., Robertson, E.J. & Efstratiadis, A. Parental imprinting of the mouse insulin-like growth factor II gene. Cell 64, (1991).
Brannan, C.I., Dees, E.C., Ingram, R.S. & Tilghman, S.M. The product of the H19 gene may function as an RNA. Molec. Cell. Biol. 10, 28–36 (1990).
Bartolomei, M.S., Zemel, S. & Tilghman, S.M. Parental imprinting of the mouse H19 gene. Nature 351, 153–155 (1991).
Pachnis, V., Brannan, C.I. & Tilghman, S.M. The structure and expression of a novel gene activated in early mouse embryogenesis. EMBO J. 7, 673–681 (1988).
Rossi, J.M. et al. Genomic analysis using a yeast artificial chromosome library with mouse DNA inserts. Proc. natn. Acad. Sci. U.S.A. 89, 2456–2460 (1992).
Rotwein, P. & Hall, L.J. Evolution of Insulin-like Growth Factor II: Characterization of the Mouse IGF-II Gene and Identification of Two Pseudo-Exons. DNA Cell Biol. 9, 725–735 (1990).
Soares, M.B. et al. Rat insulin-like growth factor II gene: a single gene with two promoters expressing a multitranscript family. J. molec. Biol. 192, 737–752 (1986).
Bell, G.I., Gerhard, D.S., Fong, N.M., Sanchez-Pescador, R. & Rall, L.B. Isolation of the human insulin-like growth factor genes: insulin-like growth factor II and insulin genes are contiguous. Proc. natn. Acad. Sci. U.S.A. 82, 6450–6454 (1985).
Riley, J. et al. A novel, rapid method for the isolation of terminal sequences from yeast artificial chromosome (YAC) clones. Nucl. Acids Res. 18, 2887–90 (1990).
Wentworth, B.M., Scharfer, I.M., Villa-Komaroff, L. & Chirgwin, J.M. Characterization of the two nonallelic genes encoding mouse preproinsulin. J. molec. Evol. 23, 305–312 (1986).
Glaser, T., Housman, D., Lewis, W.H., Gerhard, D. & Jones, C. A fine-structure deletion map of human chromosome 11p: analysis of J1 series hybrids. Somat. cell. Mol. Genet. 15, 477–501 (1989).
O'Malley, K.L. & Rotwein, P. Human tyrosine hydroxylase and insulin genes are contiguous on chromosome 11. Nucl. Acids Res. 16, 4437–4446 (1988).
Poirier, F. et al. The murine H19 gene is activated during embryonic stem cell differentiation in vitro and at the time of implantation in the developing embryo. Develop. 113, 1105–1114 (1991).
Lee, J.E., Pintar, J. & Efstratiadis, A. Pattern of the insulin-like growth factor II gene expression during early mouse embryogenesis. Develop. 110, 151–159 (1990).
Brunkow, M.E. & Tilghman, S.M. Ectopic expression of the H19 gene in mice causes prenatal lethality. Genes & Devl. 5, 1092–1101 (1991).
Bartolomei, M.S. & Tilghman, S.M. Parental imprinting of mouse chromosome 7 Sem. develop. Biol. 3, 107–117 (1992).
Nickol, J.M. & Felsenfeld, G. Bidirectional control of the chicken β- and ɛ-globin genes by a shared enhancer. Proc. natn. Acad. Sci. U.S.A. 85, 2548–2552 (1988).
Choi, O.-R.B. & Engel, J.D. Developmental regulation of β-globin switching. Cell 55, 17–26 (1988).
Zhang, Y. & Tycko, B. Monoallelic expression of the human H19 gene. Nature Genet. 1, 40–44 (1992).
Koufos, A. et al. Familial Wiedemann-Beckwith syndrome and a second Wilms tumor locus both map to 11p15.5. Am. J. hum. Genet. 44, 711–719 (1989).
Ping, A.J. et al. Genetic Linkage of Beckwith-Wiedemann syndrome to 11p15. Am. J. hum. Genet. 44, 720–723 (1989).
Niikawa, N. et al. The Wiedemann-Beckwith syndrome: pedigree studies on five families with evidence for autosomal dominant inheritance with variable expressivity. Am. J. med. Genet. 24, 41–55 (1986).
Lubinsky, M., Herrmann, J., Kosseff, A.L. & Opitz, J.M. Autosomal dominant sex-dependent transmission of the Wiedemann-Beckwith syndrome. Lancet 1, 983 (1974).
Aleck, K.A. & Hadro, T.A. Dominant inheritance of Wiedemann-Beckwith syndrome: further evidence for transmission of “unstable premutation” through carrier women. Am. J. med. Genet. 33, 155–160 (1989).
Henry, I. et al. Uniparental paternal disomy in a genetic cancer-predisposing syndrome. Nature 351, 665–667 (1991).
Turleau, C. et al. Trisomy 11p15 and Beckwith-Wiedemann syndrome: a report of two cases. Hum. Genet, 67, 219–221 (1984).
Waziri, M., Patil, S.R., Hanson, J.W. & Bartley, J.A. Abnormality of chromosome 11 in patients with features of Beckwith-Wiedemann syndrome. J. Pediatr. 102, 873–876 (1983).
Ferguson-Smith, A.C., Cattanach, B.M., Barton, S.C., Beechey, C.V. & Surani, M.A. Embryological and molecular investigations of parental imprinting on mouse chromosome 7. Nature 351, 667–670 (1991).
Carle, G.F. & Olson, M.V. An electrophoretic karyotype for yeast. Proc. natn. Acad. Sci. U.S.A. 82, 3756–3760 (1985).
Chu, G., Vollrath, D. & Davis, R.W. Separation of large DNA molecules by contour-clamped homogenous electric fields. Science 234, 1582–1585 (1986).
Church, G.M. & Gilbert, W. Genomic sequencing. Proc. natn. Acad. Sci. U.S.A. 81, 1991–1995 (1984).
Feinberg, A.P. & Vogelstein, B. A technique for radiolabeling DNA restriction fragments to high specific activity. Anal. Biochem. 137, 266–267 (1984).
Burke, D.T., Carle, G.F. & Olson, M.V. Cloning of large segments of exogenous DNA into yeast by means of artificial chromosome vectors. Science 236, 806–812 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zemel, S., Bartolomei, M. & Tilghman, S. Physical linkage of two mammalian imprinted genes, H19 and insulin–like growth factor 2. Nat Genet 2, 61–65 (1992). https://doi.org/10.1038/ng0992-61
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0992-61