Abstract
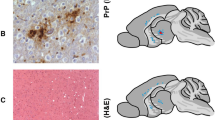
Mice were constructed carrying prion protein (PrP) transgenes with individual regions of putative secondary structure deleted. Transgenic mice with amino-terminal regions deleted remained healthy at >400 days of age, whereas those with either of carboxy-terminal α-helices deleted spontaneously developed fatal CNS illnesses similar to neuronal storage diseases. Deletion of either C-terminal helix resulted in PrP accumulation within cytoplasmic inclusions in enlarged neurons. Deletion of the penultimate C-terminal helix resulted in proliferation of rough endoplasmic reticulum. Mice with the C-terminal helix deleted were affected with nerve cell loss in the hippocampus and proliferation of smooth endoplasmic reticulum. Whether children with the human counterpart of this malady will be found remains to be determined.
Similar content being viewed by others
Article PDF
References
Brown, P. et al. Human spongiform encephalopathy: The National Institutes of Health series of 300 cases of experimentally transmitted disease. Ann. Neurol. 35, 513–529 (1994).
Gambetti, P., Parchi, P., Petersen, R.B., Chen, S.G. & Lugaresi, E. Fatal familial insomnia and familial Creutzfeldt-Jakob disease: Clinical, pathological and molecular features. Brain Pathol. 5, 43–51 (1995).
Holman, R.C., Khan, A.S., Kent, J., Strine, T.W. & Schonberger, L.B. Epidemiology of Creutzfeldt-Jakob disease in the United States, 1979–1990: Analysis of national mortality data. Neuroepidemiology 14, 174–181 (1995)
Tateishi, J. & Kitamoto, T. Inherited prion diseases and transmission to rodents. Brain Pathol. 5, 53–59 (1995).
Prusiner, S.B. in Fields Virology (eds. Fields, B.N., Knipe, D.M. & Howley, P.M.) 2901–2950 (Raven, New York, 1996).
Huang, Z. et al. Proposed three-dimensional structure for the cellular prion protein. Proc. Natl. Acad. Sci. USA 91, 7139–7143 (1994).
Schätzl, H.M., Da Costa, M., Taylor, L., Cohen, F.E. & Prusiner, S.B. Prion proteingene variation among primates. J. Mol. Biol. 245, 362–374 (1995).
Bamborough, P. et al. Prion protein structure and scrapie replication: Theoretical, spectroscopic and genetic investigations. Cold Spring Harb. Symp. Quant. Biol. 61, 495–509 (1996).
Pan, K.-M. et al. Conversion of α-helices into β-sheets features in the formation of the scrapie prion proteins. Proc. Natl. Acad. Sci. USA 90, 10962–10966 (1993).
Pergami, P., Jaffe, H. & Safar, J. Semipreparative chromatographic method to purify the normal cellular isoform of the prion protein in nondenatured form. Anal. Biochem. 236, 63–73 (1996).
Zhang, H. et al. Conformational transitions in peptides containing two putative α-helices of the prion protein. J. Mol. Biol. 250, 514–526 (1995).
Riek, R. et al. NMR structure of the mouse prion protein domain PrP(121–231). Nature 382, 180–182 (1996).
Kascsak, R.J. et al. Mouse polyclonal and monoclonal antibody to scrapie-associated fibril proteins. J. Virol. 61, 3688–3693 (1987).
Rogers, M. et al. Epitope mapping of the Syrian hamster prion protein utilizing chimeric and mutant genes in a vaccinia virus expression system. J. Immunol. 147, 3568–3574 (1991).
Scott, M.R., Köhler, R., Foster, D. & Prusiner, S.B. Chimeric prion protein expression in cultured cells and transgenic mice. Protein Sci. 1, 986–997 (1992).
Scott, M. et al. Propagation of prions with artificial properties in transgenic mice expressing chimeric PrP genes. Cell 73, 979–988 (1993).
Rogers, M., Yehiely, F., Scott, M. & Prusiner, S.B. Conversion of truncated and elongated prion proteins into the scrapie isoform in cultured cells. Proc. Natl. Acad. Sci. USA 90, 3182–3186 (1993).
Fischer, M. et al. Prion protein (PrP) with amino-proximal deletions restoring susceptibility of PrP knockout mice to scrapie. EMBO J. 15, 1255–1264 (1996).
Muramoto, T., Scott, M., Cohen, F. & Prusiner, S.B. Recombinant scrapie-like prion protein of 106 amino acids is soluble. Proc. Natl. Acad. Sci. USA 93, 15457–15462 (1996).
Barry, R.A. & Prusiner, S.B. Monoclonal antibodies to the cellular and scrapie prion proteins. J. Infect. Dis. 154, 518–521 (1986).
Turk, E., Teplow, D.B., Hood, L.E. & Prusiner, S.B. Purification and properties of the ellular and scrapie hamster prion proteins. Eur. J. Biochem. 176, 21–30 (1988).
Huang, Z., Prusiner, S.B. & Cohen, F.E. Scrapie prions: A three-dimensional model of an infectious fragment. Folding Design 1, 13–19 (1996).
Büeler, H. et al. Normal development and behaviour of mice lacking the neuronal cell-surface PrP protein. Nature 356, 577–582 (1992).
Phillips, M.J., Poucell, S. & Valencia, P. The Liver: An Atlas and Text of Ultrastructural Pathology. (Raven, New York, 1987).
Kitamoto, T., lizuka, R. & Tateishi, J. An amber mutation of prion protein in Gerstmann-Straussler syndrome with mutant PrP plaques. Biochem. Biophys. Res. Commun. 192, 525–531 (1993).
Ghetti, B. et al. Vascular variant of prion protein cerebral amyloidosis with τpositive neurofibrillary tangles: The phenotype of the stop codon 145 mutation in PRNP. Proc. Natl. Acad. Sci. USA 93, 744–748 (1996).
Meyer, R.K. et al. Separation and properties of cellular and scrapie prion proteins. Proc. Natl. Acad. Sci. USA 83, 2310–2314 (1986).
Tatzelt, J., Prusiner, S.B. & Welch, W. Chemical chaperones interfere with the formation of scrapie prion protein. EMBO J. 15, 6363–6373 (1996).
Gossen, M. et al. Transcriptional activation by tetracyclines in mammalian cells. Science 268, 1766–1769 (1995).
No, D., Yao, T.-P. & Evans, R.M. Ecdysone-inducible gene expression in mammalian cells and transgenic mice. Proc. Natl. Acad. Sci. USA 93, 3346–3351 (1996).
Mehlhorn, I. et al. High-level expression and characterization of a purified 142-residue polypeptide of the prion protein. Biochemistry 35, 5528–5537 (1996).
Gabizon, R. et al. Insoluble wild-type and protease-resistant mutant prion protein in brains of patients with inherited prion disease. Nature Med. 2, 59–64 (1996).
Rogers, M., Taraboulos, A., Scott, M., Groth, D. & Prusiner, S.B. Intracellular accumulation of the cellular prion protein after mutagenesis of its Asn-linked glycosylation sites. Glycobiology 1, 101–109 (1990).
Taraboulos, A. et al. Acquisition of protease resistance by prion proteins in scrapie-infected cells does not require asparagine-linked glycosylation. Proc. Natl. Acad. Sci. USA 87, 8262–8266 (1990).
Munro, S. & Pelham, H.R. A C-terminal signal prevents secretion of luminal ER proteins. Cell 48, 899–907 (1987).
Pelham, H.R.B. The control of protein exit from the endoplasmic reticulum. Annu. Rev. Cell Biol. 5, 1–23 (1989).
Scott, M. et al. Transgenic mice expressing hamster prion protein produce species specific scrapie infectivity and amyloid plaques. Cell 59, 847–857 (1989).
Kitamoto, T. et al. Abnormal isoform of prion proteins accumulates in the synaptic structures of the central nervous system in patients with Creutzfeldt-Jakob disease. Am. J. Pathol. 140, 1285–1294 (1992).
Taraboulos, A. et al. Regional mapping of prion proteins in brains. Proc. Natl. Acad. Sci. USA 89, 7620–7624 (1992).
Hsiao, K.K. et al. Serial transmission in rodents of neurodegeneration from transgenic mice expressing mutant prion protein. Proc. Natl. Acad. Sci. USA 91, 9126–9130 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Muramoto, T., DeArmond, S., Scott, M. et al. Heritable disorder resembling neuronal storage disease in mice expressing prion protein with deletion of an α-helix. Nat Med 3, 750–755 (1997). https://doi.org/10.1038/nm0797-750
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0797-750
This article is cited by
-
Secretory pathway retention of mutant prion protein induces p38-MAPK activation and lethal disease in mice
Scientific Reports (2016)
-
Intracerebral Infusion of Antisense Oligonucleotides Into Prion-infected Mice
Molecular Therapy - Nucleic Acids (2012)
-
Conserved properties of human and bovine prion strains on transmission to guinea pigs
Laboratory Investigation (2011)
-
Asymptomatic deer excrete infectious prions in faeces
Nature (2009)
-
The two faces of protein misfolding: gain- and loss-of-function in neurodegenerative diseases
The EMBO Journal (2008)