Abstract
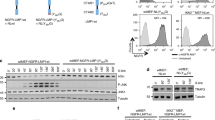
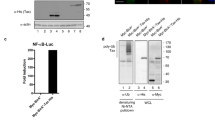
Expression of the oncogenic Epstein-Barr virus (EBV)-encoded Latent Membrane Protein 1 (LMP1) activates signalling on the NF-κB axis through two distinct domains in the cytoplasmic C-terminus of the protein, namely CTAR1 (aa 187–231) and CTAR2 (aa 351–386). Whilst this effect is responsible for some of the functional consequences of LMP1 expression, additional LMP1-mediated signalling pathways may exist which contribute to the pleiotropic activities of this protein. In this study we provide evidence of a kinase cascade being activated by LMP1. Thus, we demonstrate that stable or transient expression of the LMP1 prototype from B95.8 in cells of epithelial or B cell origin activates the c-Jun N-terminal kinase (JNK, also known as the stress-activated protein kinase, SAPK) pathway, an effect which was found to be mediated through CTAR2 but not CTAR1. LMP1 from the Cao viral strain or LMP1 homologues from the simian EBV naturally infecting baboons and rhesus monkeys were also able to activate JNK. This phenomenon translates to induction of AP-1, a transcription factor which is readily activated by growth factors and mitogens. Interestingly, an LMP1/CD40 chimaera comprising of the N-terminus and transmembrane domain of LMP1 and the cytoplasmic tail of CD40 which shares a common TRAF binding motif with CTAR1, effectively induced JNK. As NF-κB and JNK are co-activated in LMP1-expressing cells, we investigated whether the two pathways are overlapping or independent. We have found that inhibition of NF-κB by metabolic inhibitors or a constitutively active mutated IκBα does not impair the ability of LMP1 to signal on the JNK axis. Conversely, whilst a dominant negative mutated SEK (JNKK) inhibited LMP1-induced JNK activation, it did not affect NF-κB suggesting that these two LMP1-mediated pathways are divergent.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eliopoulos, A., Young, L. Activation of the cJun N-terminal kinase (JNK) pathway by the Epstein-Barr virus-encoded latent membrane protein 1 (LMP1). Oncogene 16, 1731–1742 (1998). https://doi.org/10.1038/sj.onc.1201694
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1201694
Keywords
This article is cited by
-
DUSP16 promotes cancer chemoresistance through regulation of mitochondria-mediated cell death
Nature Communications (2021)
-
CRISPR/Cas9-mediated LMP1 knockout inhibits Epstein-Barr virus infection and nasopharyngeal carcinoma cell growth
Infectious Agents and Cancer (2019)
-
Molecular mechanisms of EBV-driven cell cycle progression and oncogenesis
Medical Microbiology and Immunology (2019)